 ※ Our History
The company core team is made up of 15 + years of experience in medical equipment research and development, production, sales, product use and service of senior experts, at present has developed four series (digital color doppler ultrasound in the diagnosis of series, a series of ultrasonic doppler in the diagnosis of electrocardiogram machine series, the series of patient monitor), 20 of the distinctive product, at present already obtained the TUV rheinland CE certification, all products by the guangdong medical equipment quality supervision and inspection from the listed testing, in China in December 2019, the CFDA registration certificate of medical equipment.
※ Our Factory
The company has a strategic partnership with the department of medical bioengineering of Shenzhen university, so the company's main research and development base is located in Shenzhen university. The factory is located in shenzhen, the pilot demonstration zone of China's reform and opening up. At present, the main assembly and inspection workshop of the factory is set up in yinlong industrial city, longgang district, shenzhen. It covers an area of 1000 square meters and has 30 senior technical workers.
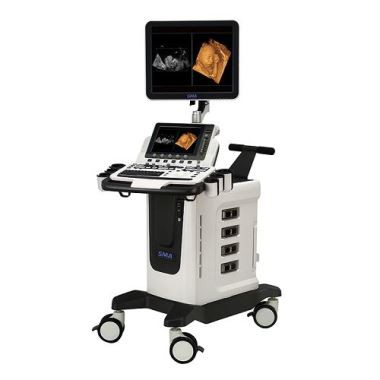
※ Our Product
1. Full digital color ultrasonic doppler diagnosis series,
2. Ultrasonic doppler diagnosis series,
3. Electrocardiogram machine series,
4. Patient monitor series
5. Medical software series
6. Various medical equipment and consumables

※ Product Application
It covers large, medium and small medical institutions with specialized ultrasound examination, ordinary bedside, outpatient, emergency and physical examination, general department and electrocardiogram examination, ICU, anesthesiology, emergency and bedside patient monitoring
※ Our Certificate
CE certificate and 5 software Copyrights
※ Production Equipment
S/N |
Device ID |
Device Name |
Specifications/ Model |
1 |
TZ-001 |
Electric torque batch |
BY800 |
2 |
TZ-002 |
Blood flow mode |
|
3 |
TZ-003 |
Temperature rise monitor |
|
4 |
TZ-004 |
Blood pressure tester |
|
5 |
NL-001 |
Torque tester |
HP-20 |
6 |
JJ-001 |
Electrostatic ring tester |
498 |
7 |
ZK-001 |
Medical grounding resistance tester |
RK2678YM |
8 |
LD001 |
Medical leakage current tester |
RK2675Y |
9 |
NY001 |
Pressure tester |
RK670Y |
10 |
TM001 |
High frequency body model |
KS107BG |
11 |
TM002 |
Low frequency body model |
KS107BD |
12 |
TM003 |
Sliced body model |
KS107BQ |
13 |
LT-001 |
Soldering iron |
BY8010 |
14 |
LT-002 |
4D body model |
BY8010 |
15 |
JJ-002 |
Electric iron temperature point tester |
191 |
16 |
JQ-001 |
Glue gun |
80-100W |
17 |
WD-001 |
Temperature hygrometer |
MBS-325 |
18 |
WD-002 |
Simulated blood oxygen detector |
MBS-325 |
19 |
WD-003 |
High temperature test environment |
|
20 |
MK-TJD-001 |
Intelligent electrocardiogram machine - verification instrument for heart and brain monitor |
ECG-1C |
21 |
MK-TMN-001 |
Various abnormal ecg simulators |
WDCS-048 |
※ Production Market
Africa region: The company had set up a factory in Africa and become the first medical equipment manufacturer with independent intellectual property rights in Africa. The products have been recognized by many African countries and governments and signed long-term cooperation agreements with annual sales of more than 2 million US dollars
Southeast Asia region: The product has been registered in Indonesia, annual sales of more than 1 million US dollars
Central Asia region: Actively developing the central Asia market, with annual sales up to US $200,000
Arab region: Developing mature agency distribution channels with annual sales of $300,000
South America region: Actively developing the central Asian market, with annual sales up to $300,000
※ Our Service
Pre-sales: Provide comprehensive product knowledge explanation, prototype trial and trainingIn
Sale: Collect trial information, complete customer customization and accurate delivery
After-sales service: 24-hour professional after-sales service
|

